
(01)
FLOWSENSE
Role:
User Experience
Interaction Design
Industrial Design
Team:
Research & Development at Rhaeos
Year:
2024 - 2025
(01)
FLOWSENSE

(01)
FLOWSENSE
Role:
User Experience
Interaction Design
Industrial Design
Team:
Research & Development at Rhaeos
Year:
2024 - 2025
(01)
FLOWSENSE
Brief
How might we empower Hydrocephalus patients to effectively monitor their shunt health and share data with neurologists, while ensuring device placement and symptom tracking?
Brief
How might we empower Hydrocephalus patients to effectively monitor their shunt health and share data with neurologists, while ensuring device placement and symptom tracking?
Brief
How might we empower Hydrocephalus patients to effectively monitor their shunt health and share data with neurologists, while ensuring device placement and symptom tracking?
Context
Context
Shunt malfunction in hydrocephalus patients is challenging to monitor with traditional methods.
Shunt malfunction in hydrocephalus patients is challenging to monitor with traditional methods.
1M
1M
70%
70%
16%
16%
Americans living with Hydrocephalus
Americans living with Hydrocephalus
Americans living with Hydrocephalus
of the patients are discharged the same day after visiting the ED for symptoms of Shunt Malfunction
of the patients are discharged the same day after visiting the ED for symptoms of Shunt Malfunction
Patients get readmitted within 30 days after shunt surgery
Patients get readmitted within 30 days after shunt surgery
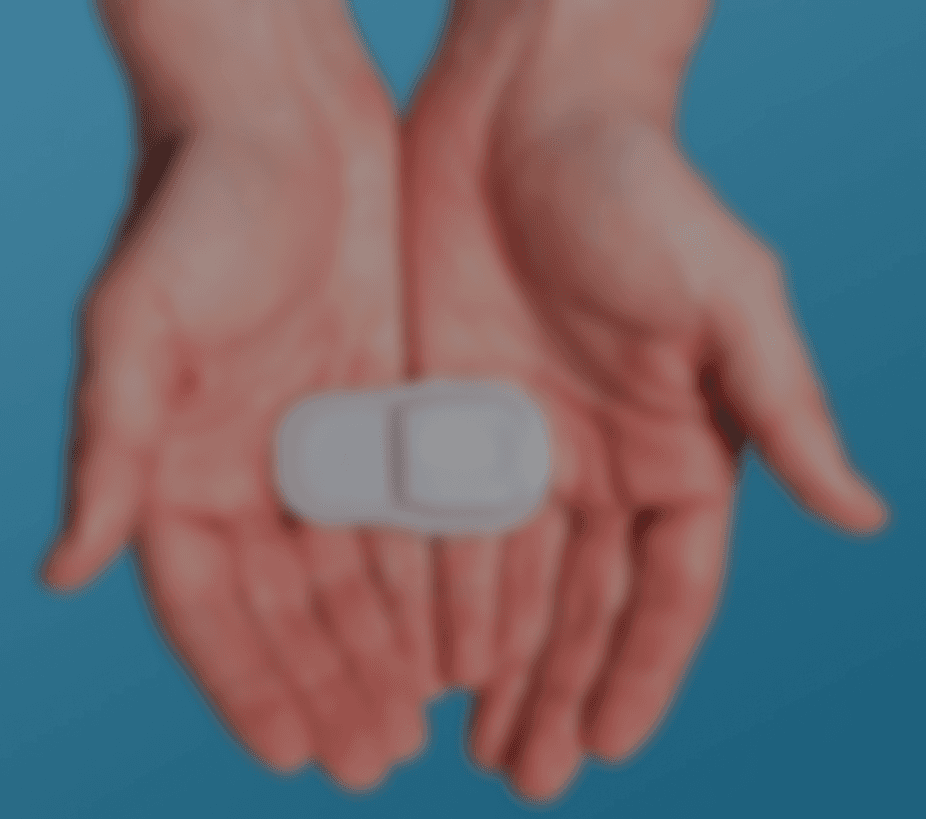
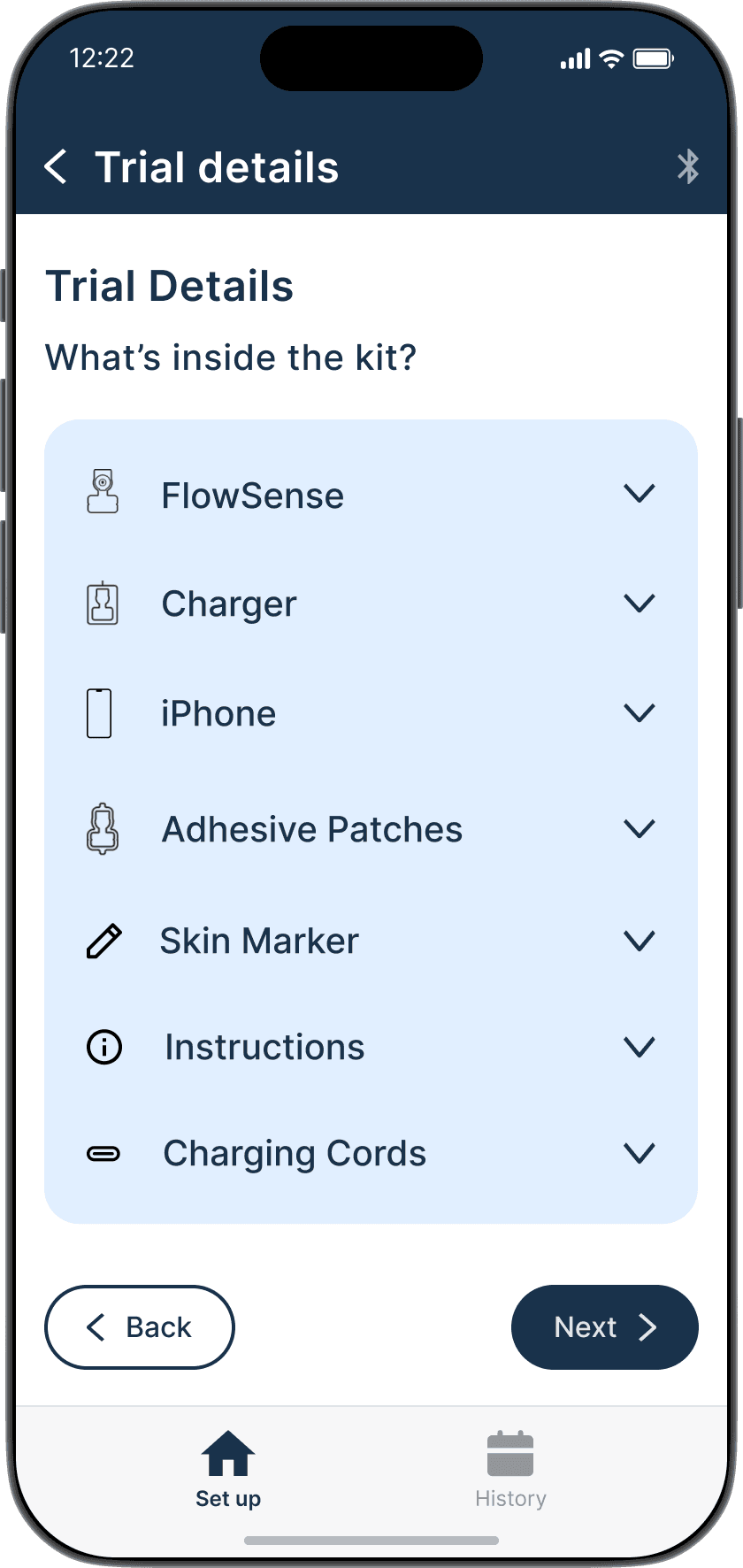
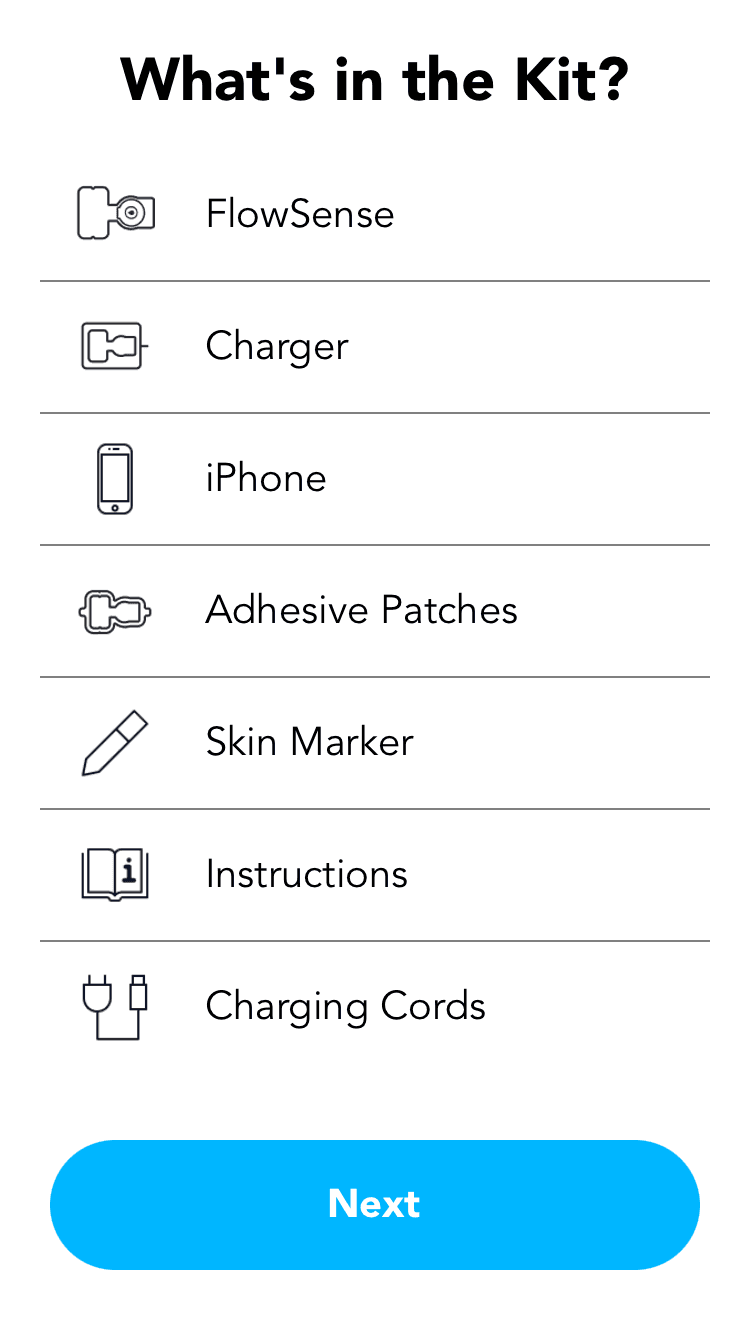
FlowSense is a wireless, noninvasive system with a thermal flow sensor that adheres to the patient's chest over a shunt to detect CSF (Cerebrospinal Fluid) flow presence and magnitude.
Soft and bandage-sized, it transmits data wirelessly to a custom mobile app.
FlowSense enables real-time shunt monitoring in clinics, hospitals, emergency departments, and even at patients' homes reducing imaging, hospital stays, and readmission costs.
FlowSense is a system with a wireless, noninvasive thermal flow sensor that adheres to the patient's neck over a shunt to detect CSF (Cerebrospinal Fluid) flow presence and magnitude.
Soft and bandage-sized, it transmits data wirelessly to a custom mobile app.
FlowSense enables real-time shunt monitoring in clinics, hospitals, emergency departments, and even at patients' homes reducing unnecessary imaging, hospital stays, and readmission costs.
FlowSense Gen 1: Hospital use
FlowSense Gen 1: Hospital use
Challenges
Challenges
Even with a non-invasive wearable, patients may struggle with device set up, accurate placement, and symptom tracking at home.
Device set up, accurate placement, and symptom tracking at home.
Phase 01: Design
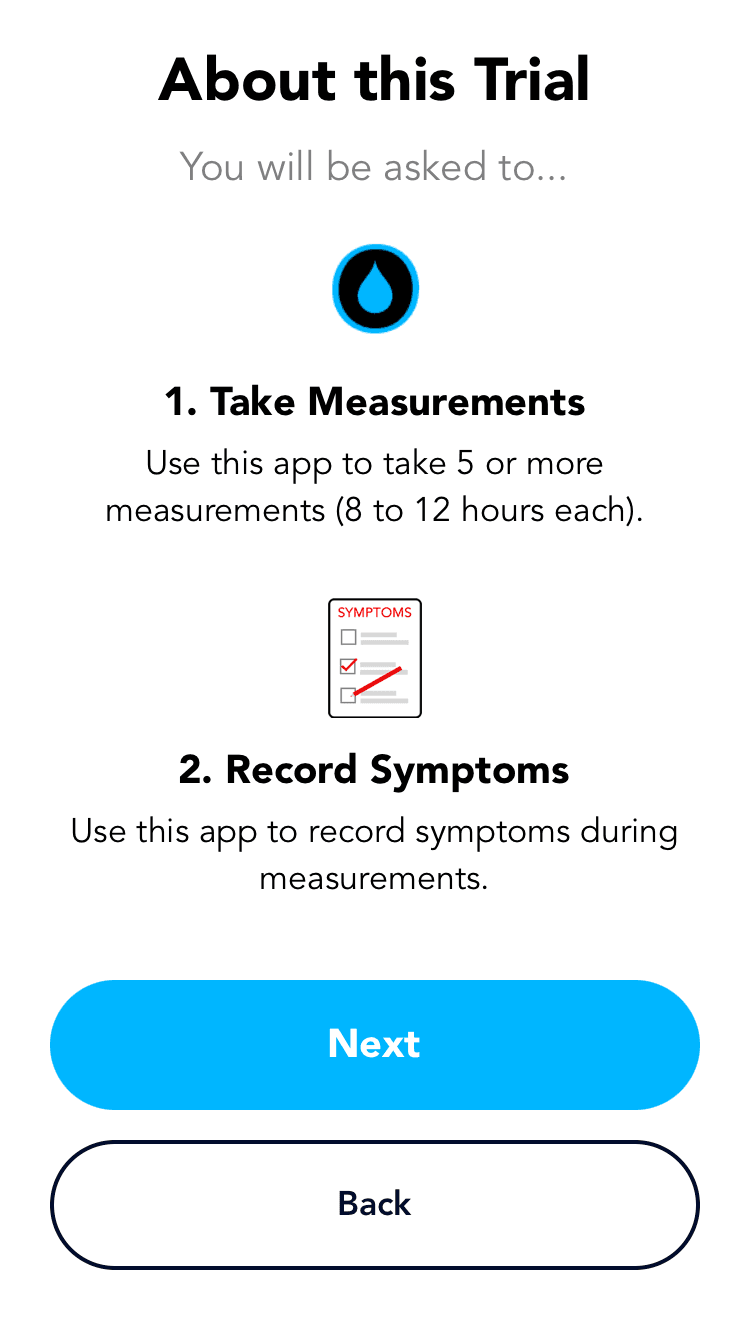
Flowsense app is a companion that works in sync with the device, guiding the patients to independently manage shunt health with confidence.
Flowsense app is a companion that works in sync with the device, guiding the patients to independently manage shunt health with confidence.


FlowSense Gen 2 Device: Focused towards home use


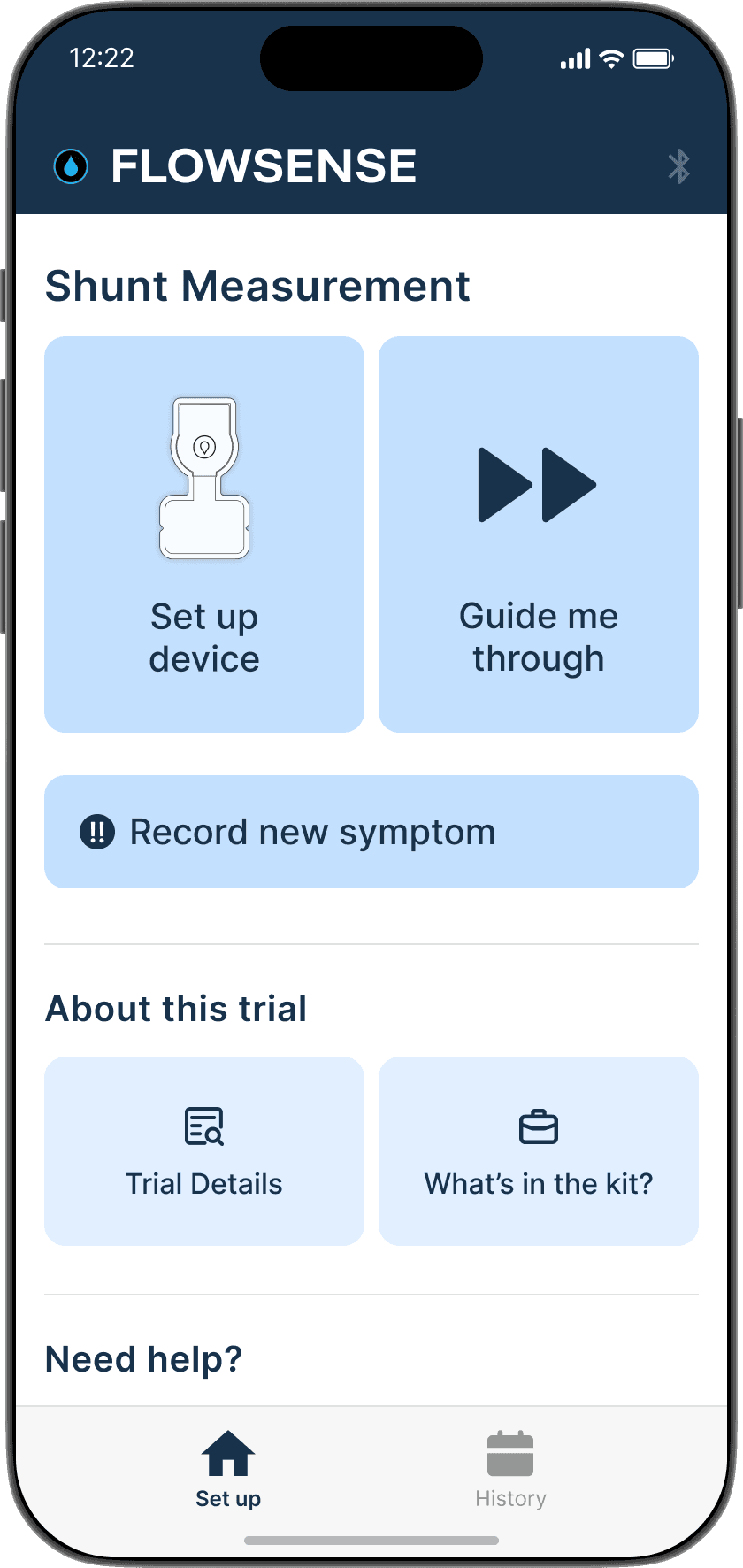





FlowSense App: Provides patients with easy access to a seamless monitoring experience
Phase 01: Design
Flowsense app is a companion that works in sync with the device, guiding the patients to independently manage shunt health with confidence.

FlowSense Gen 2 Device: Focused towards home use



FlowSense App: Provides patients with easy access to a seamless monitoring experience
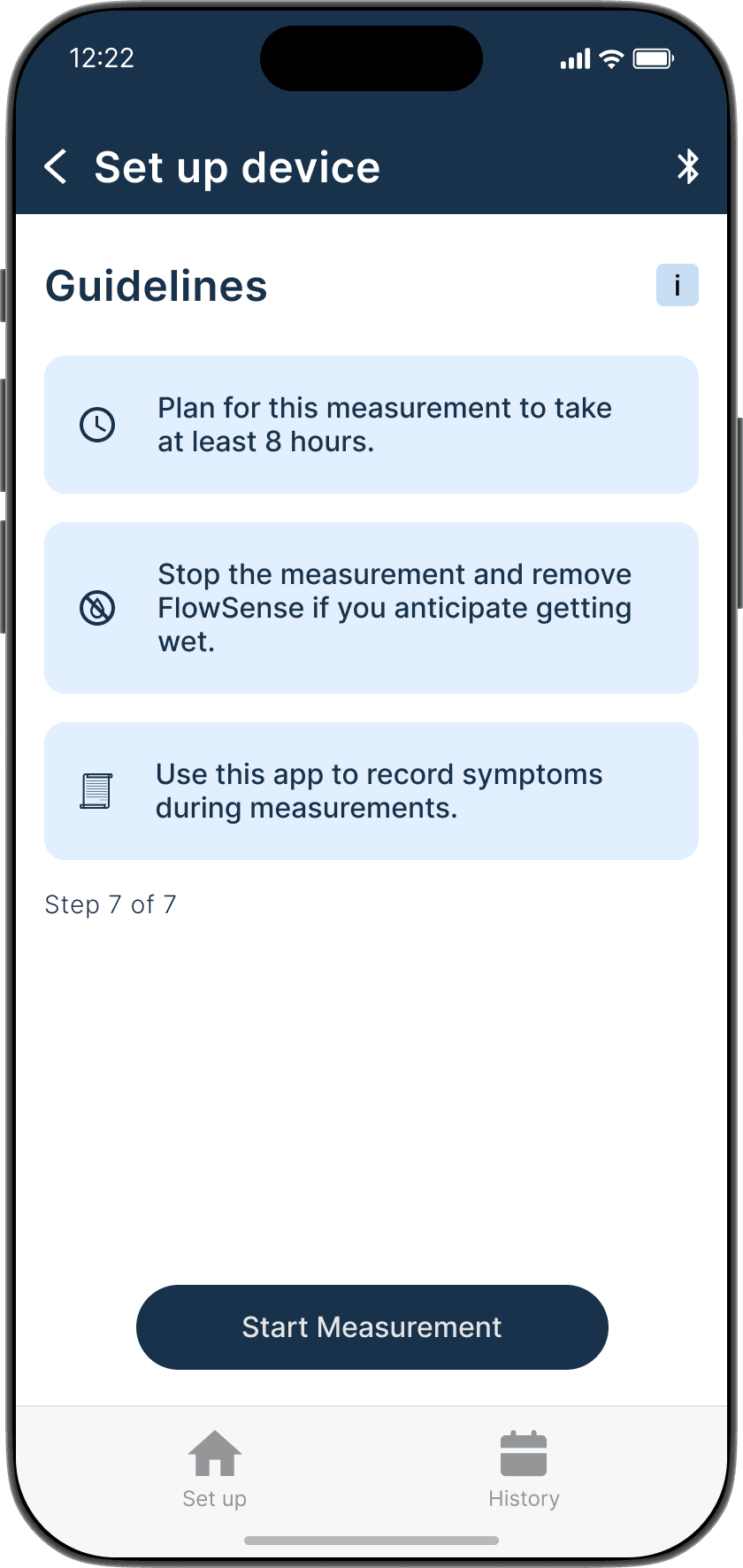
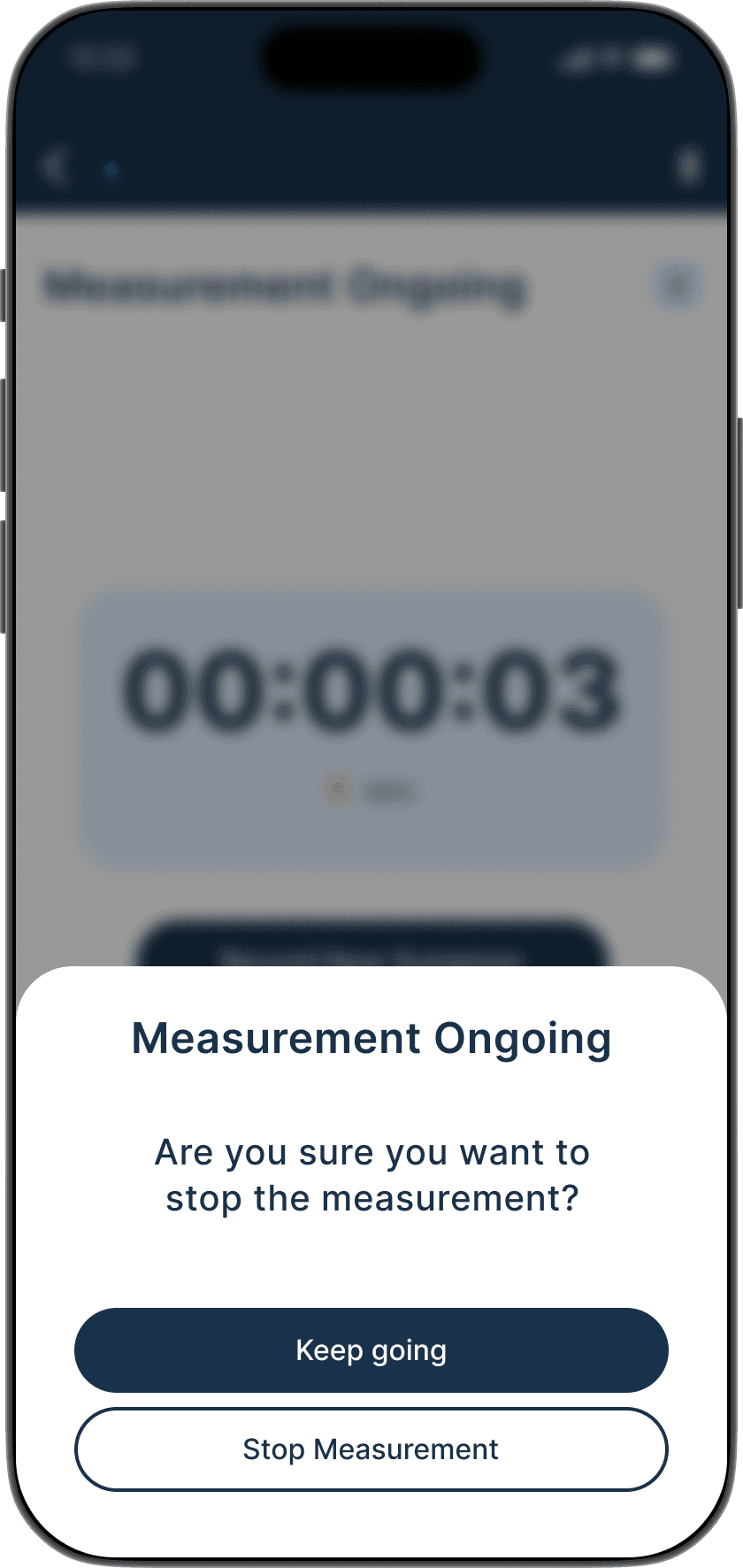
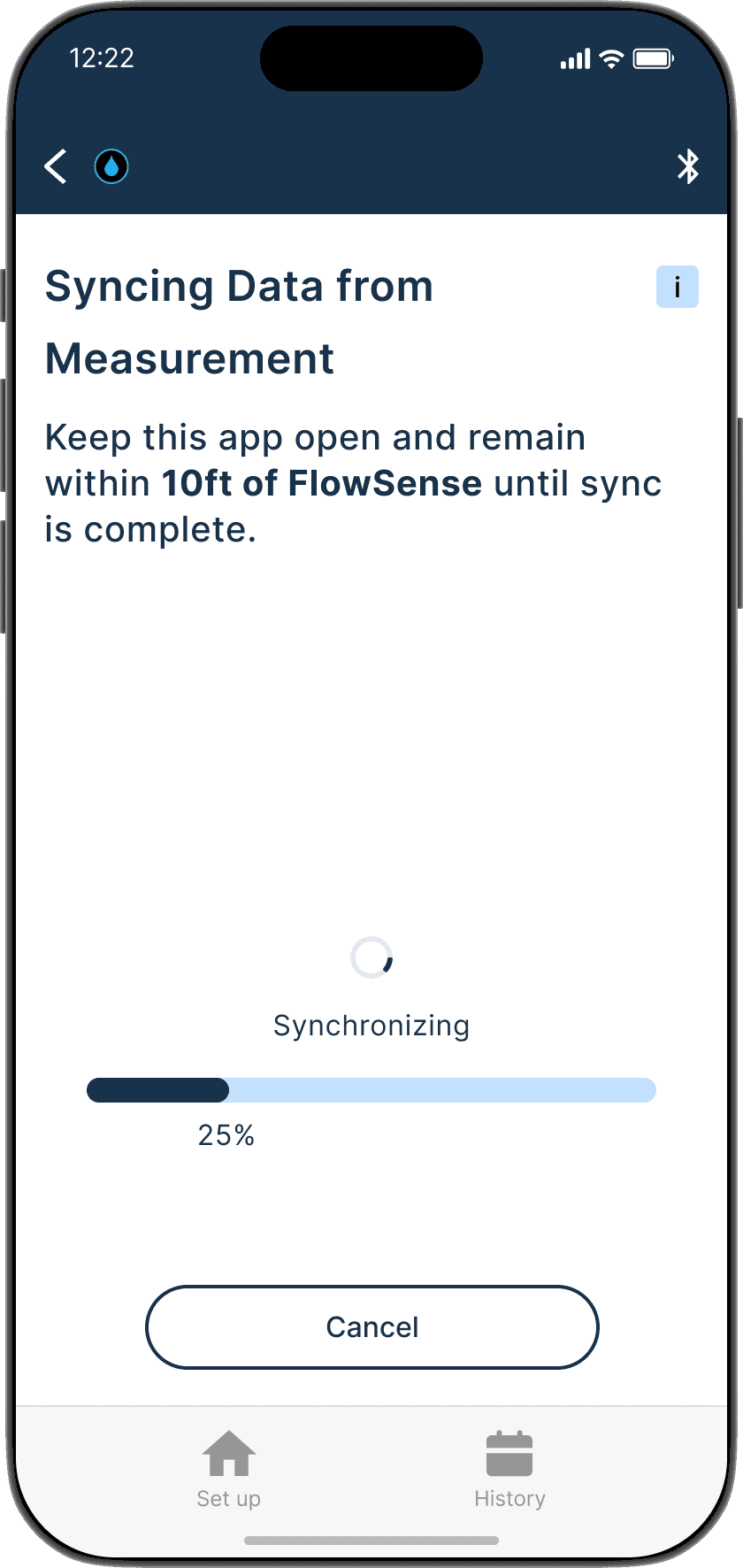
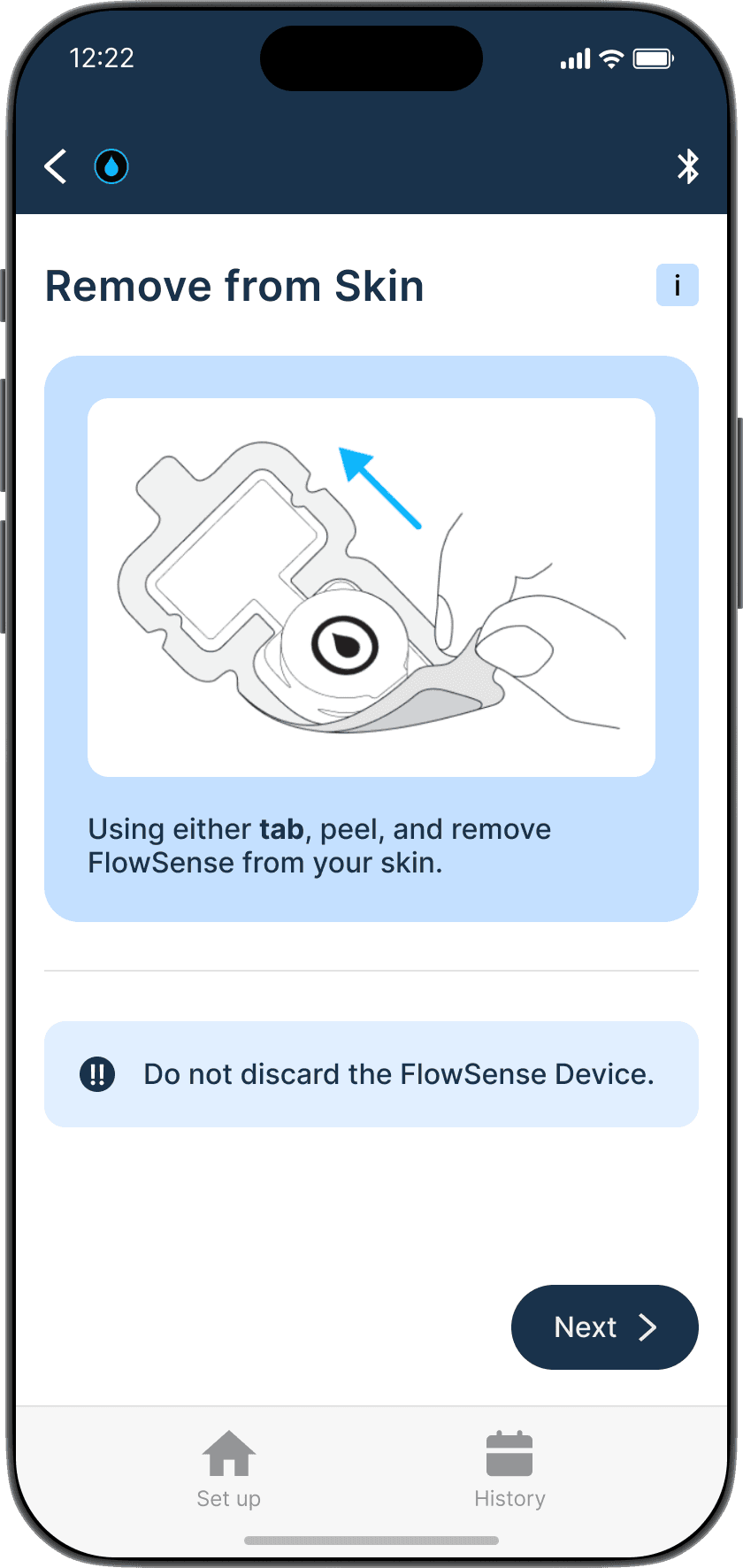
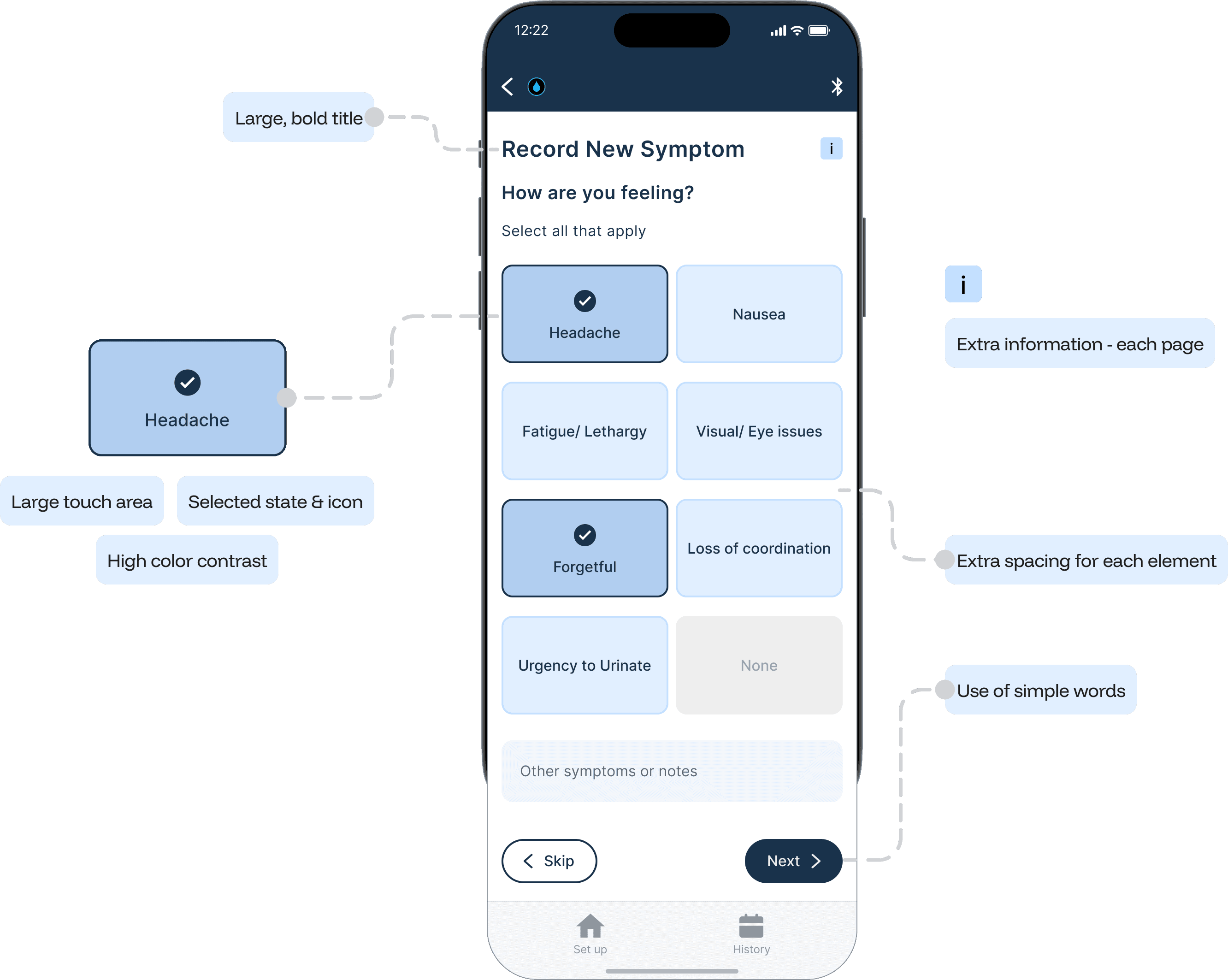
The Guide feature provides a step-by-step interactive walkthrough, reducing cognitive load for first time users and those who might need assistance, ensuring accurate set up and measurements every time.
The Guide feature provides a step-by-step interactive walkthrough, reducing cognitive load for first time users and those who might need assistance, ensuring accurate set up and measurements every time.
The Guide feature provides a step-by-step interactive walkthrough, reducing cognitive load for first time users and those who might need assistance, ensuring accurate set up and measurements every time.
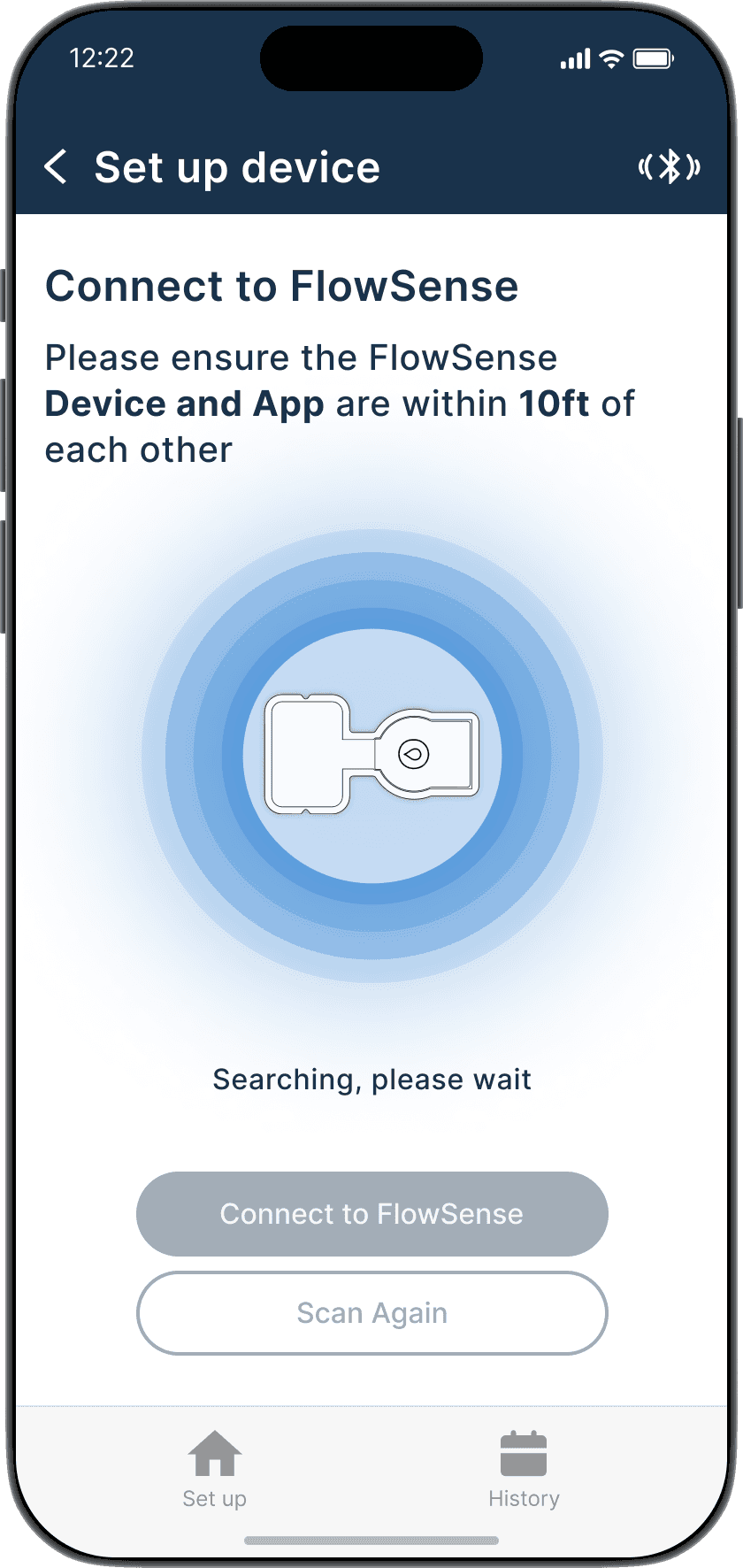



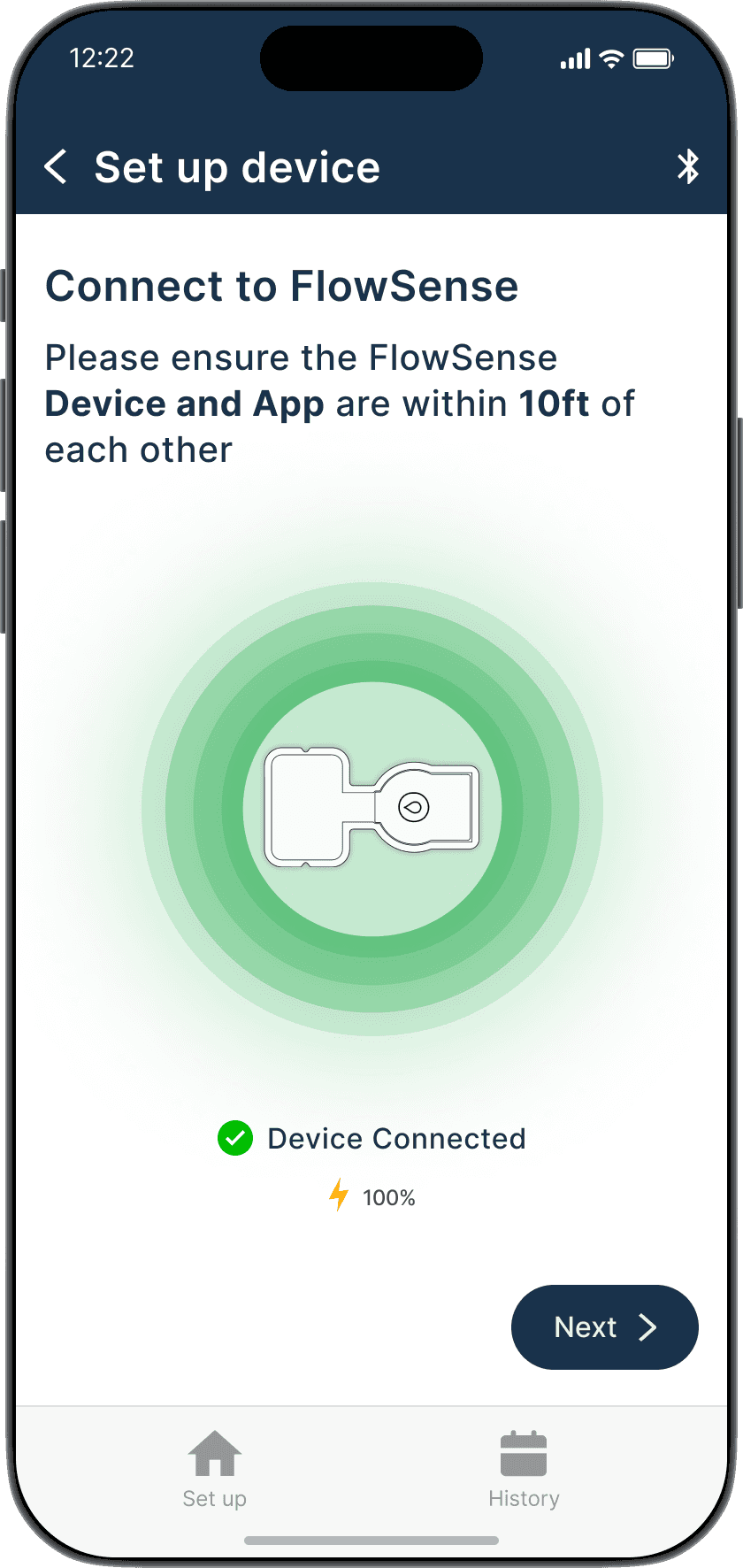
Bluetooth pairing with FlowSense device
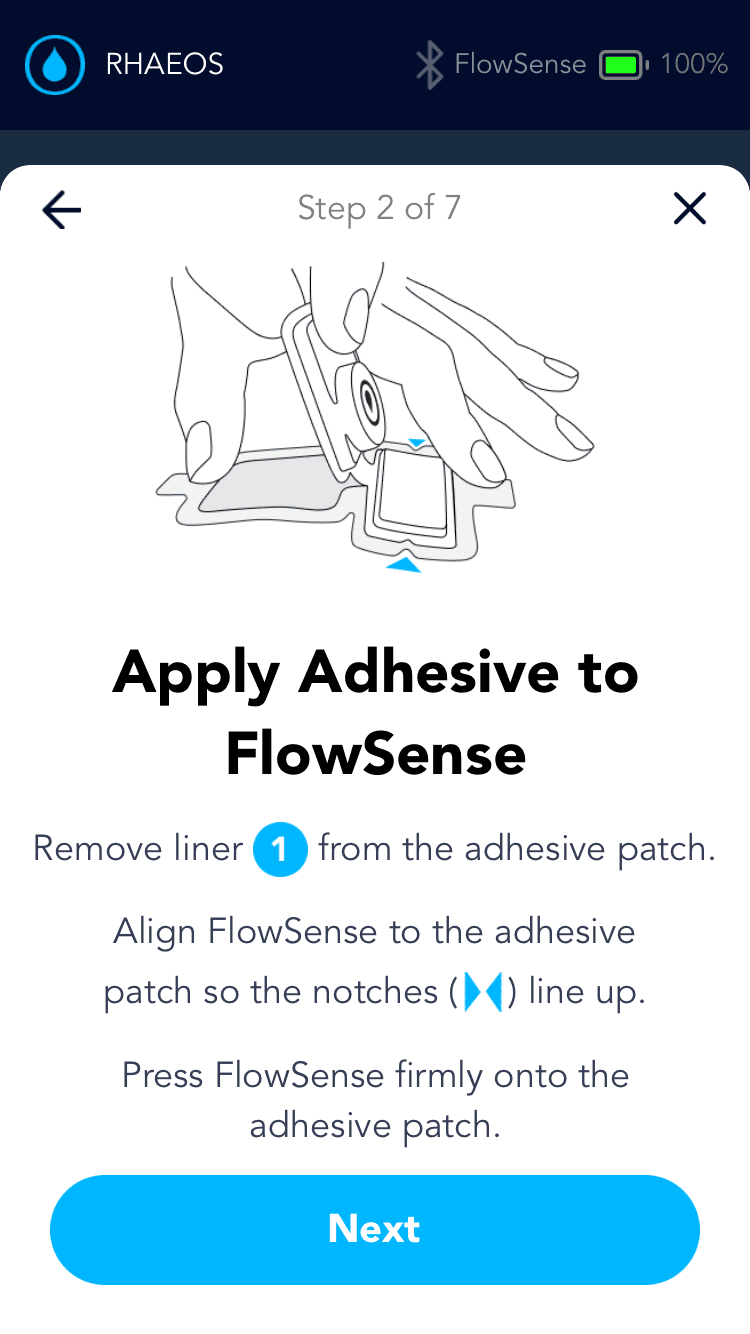
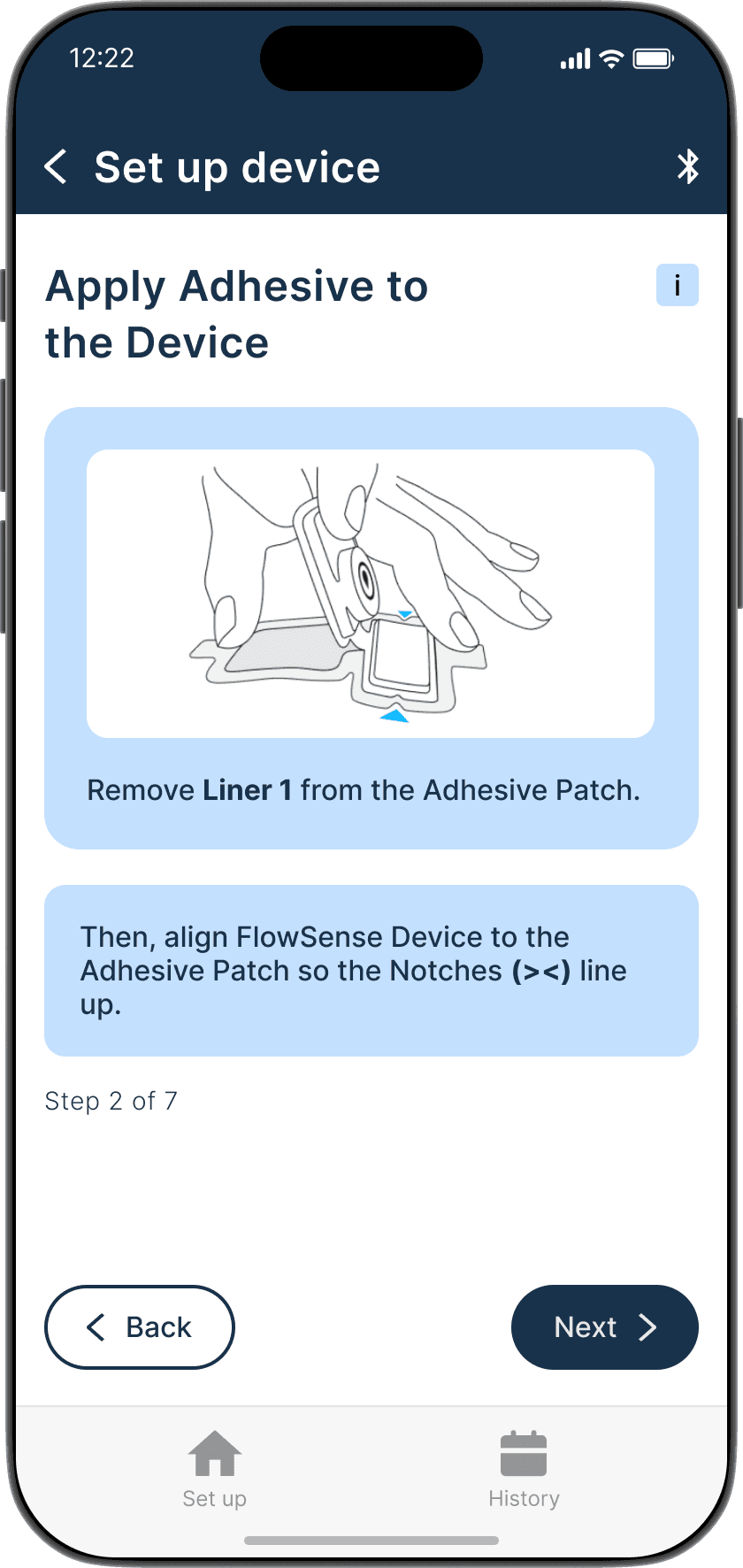
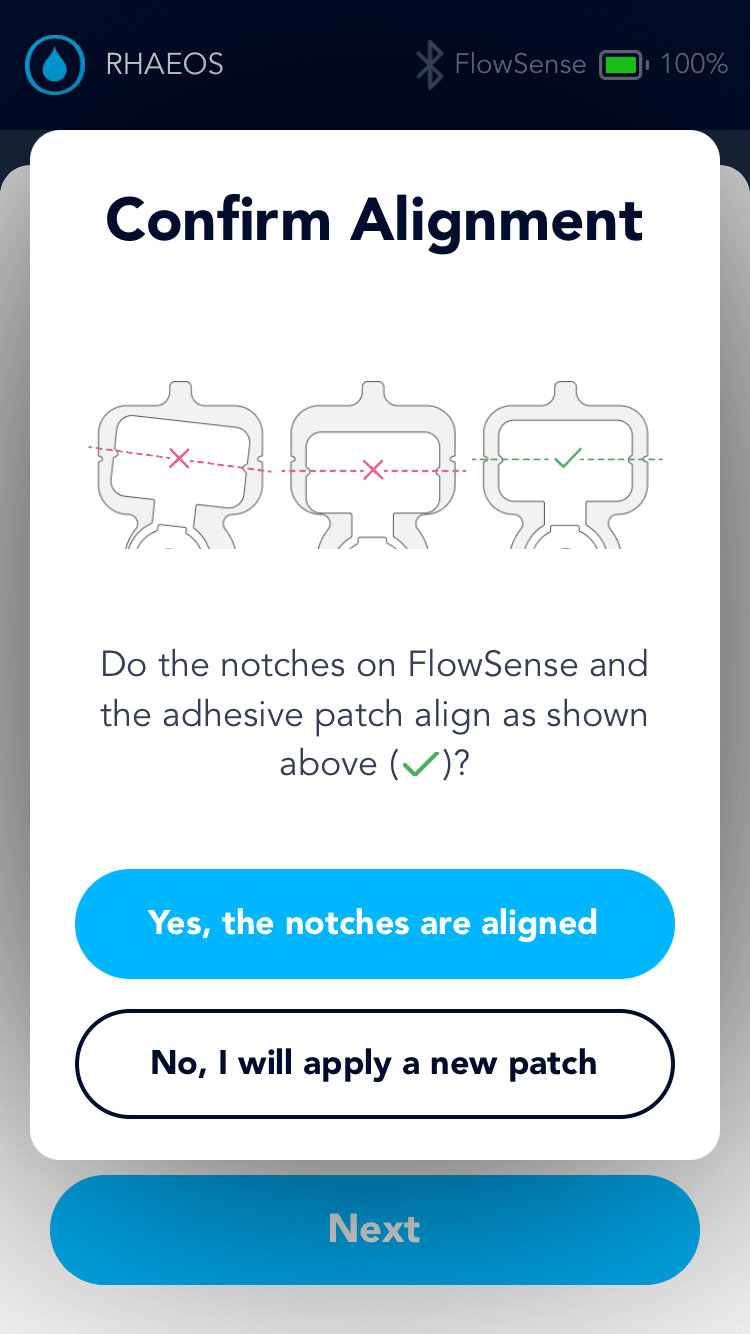
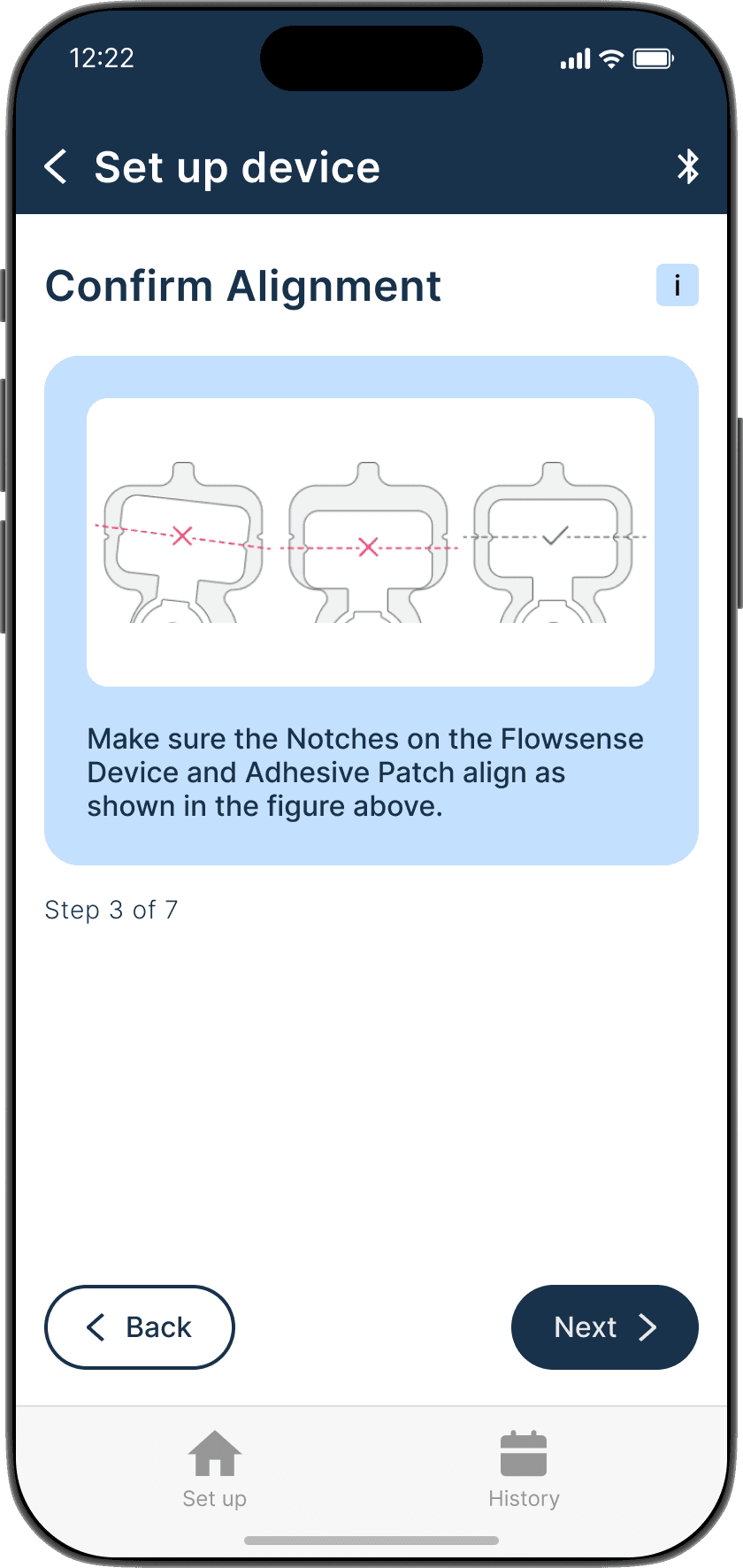
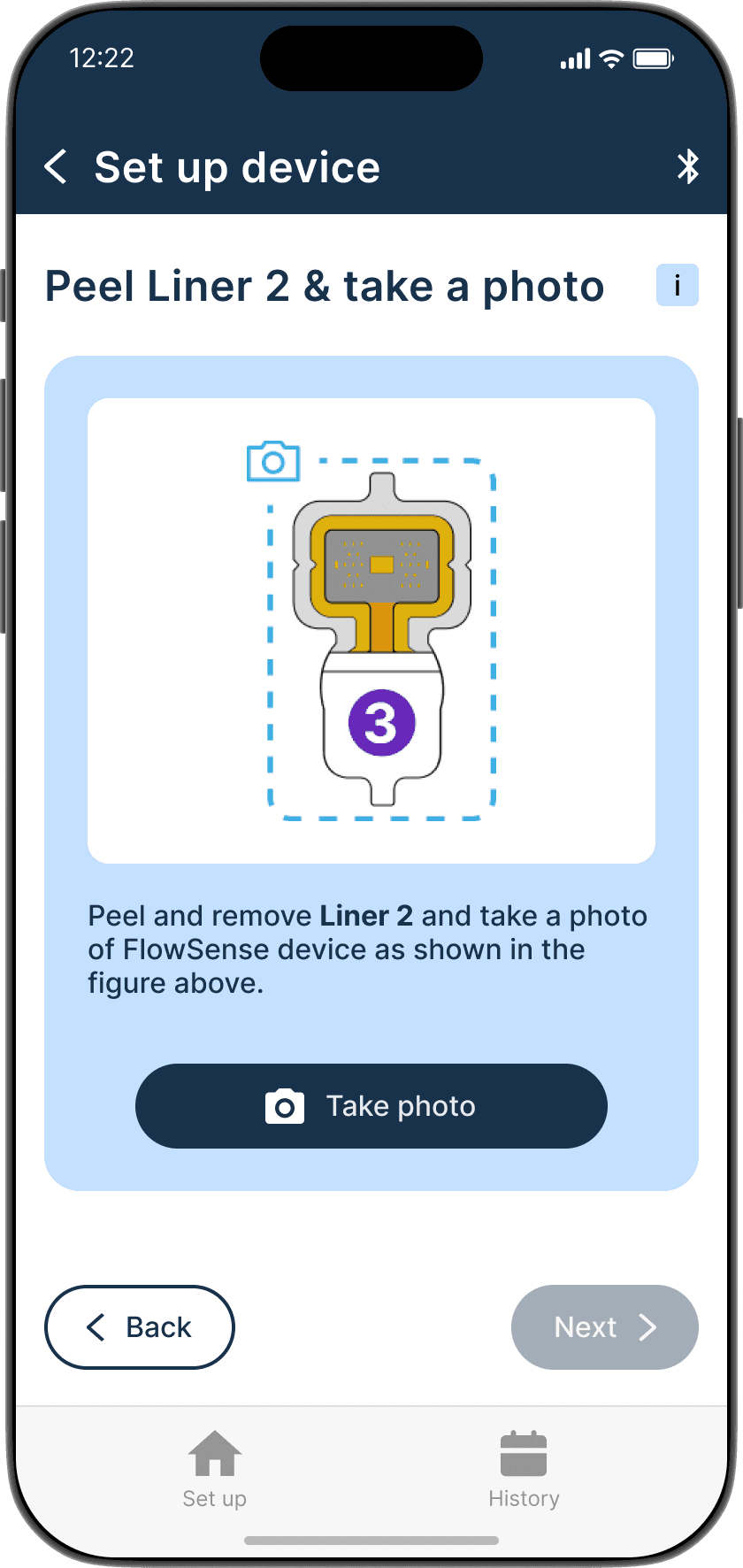
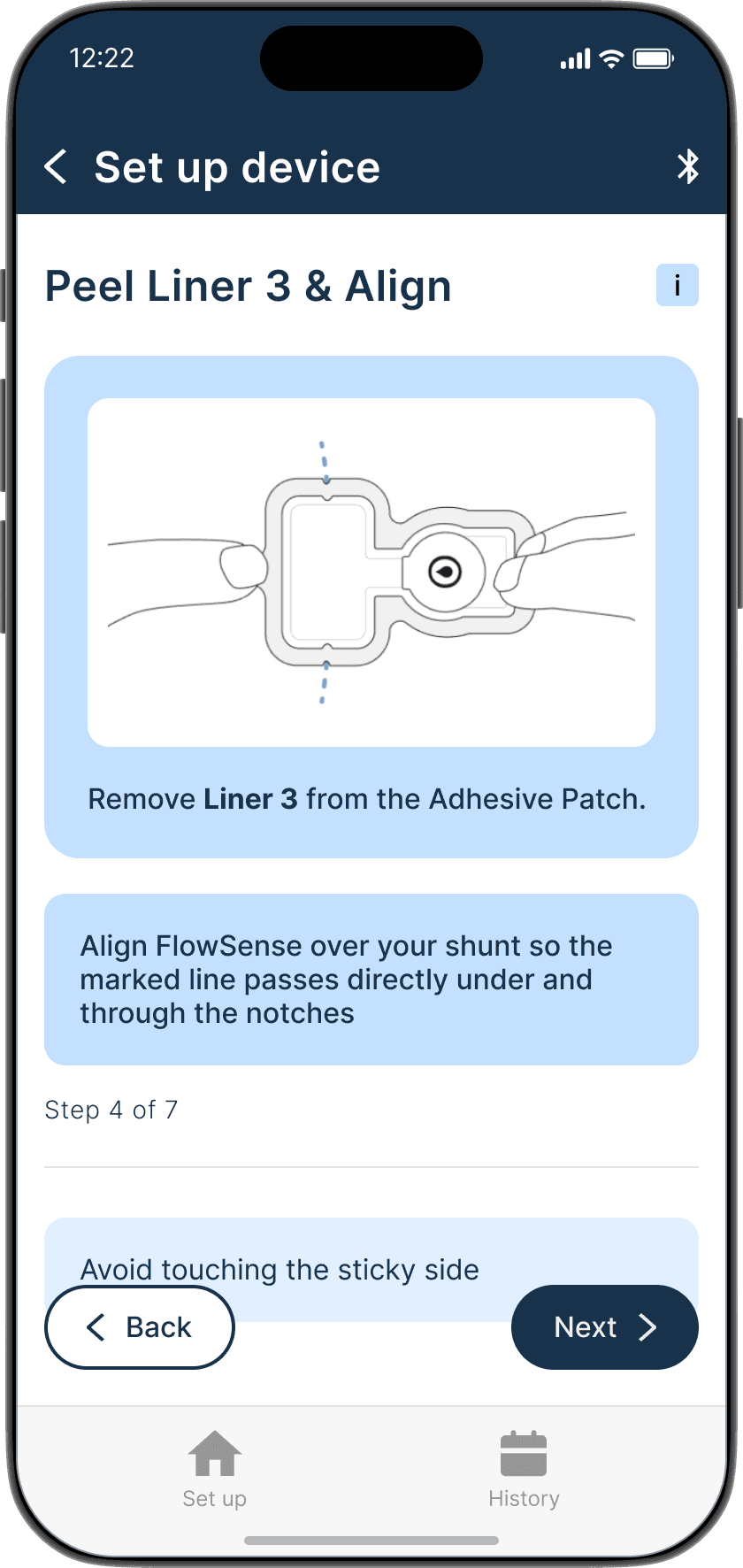
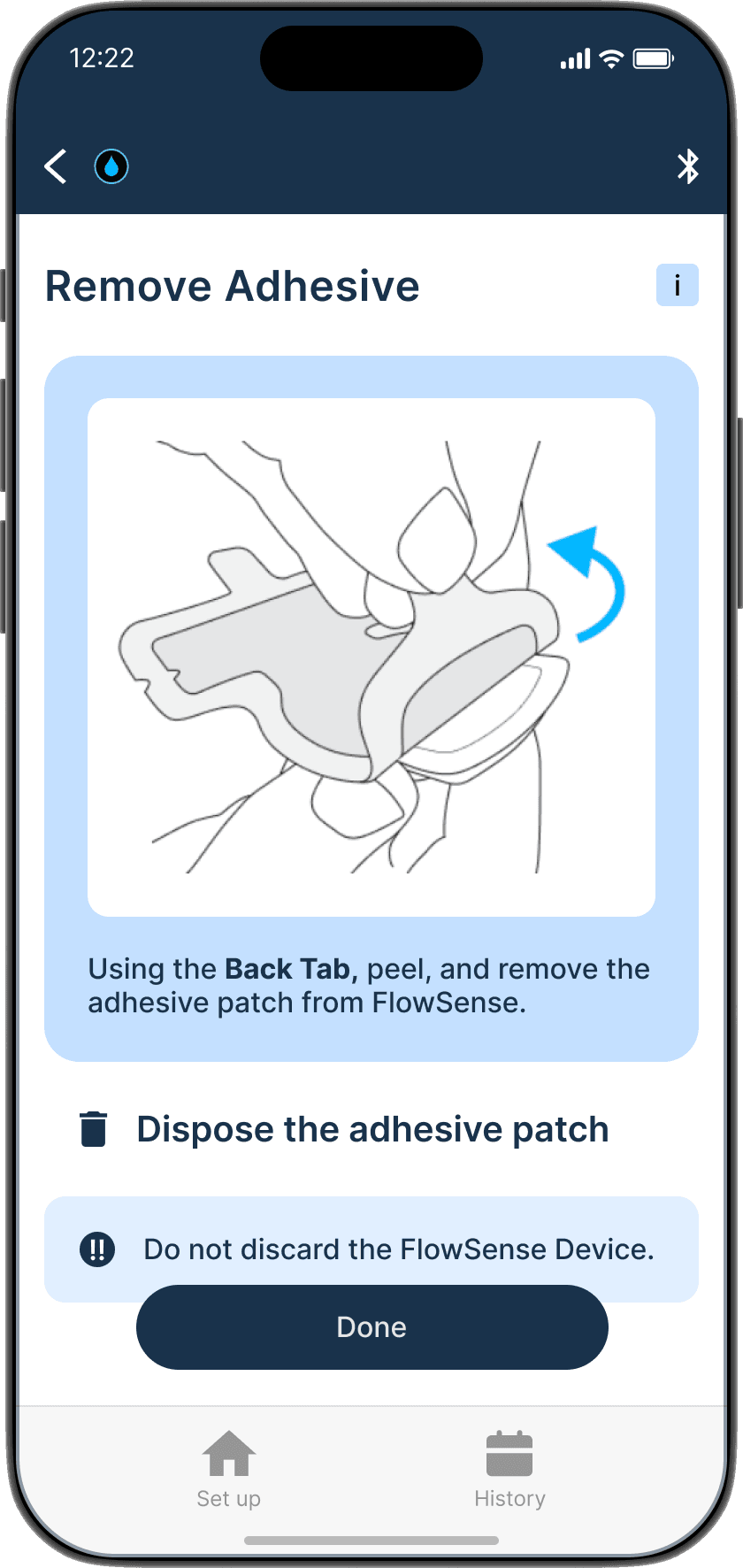
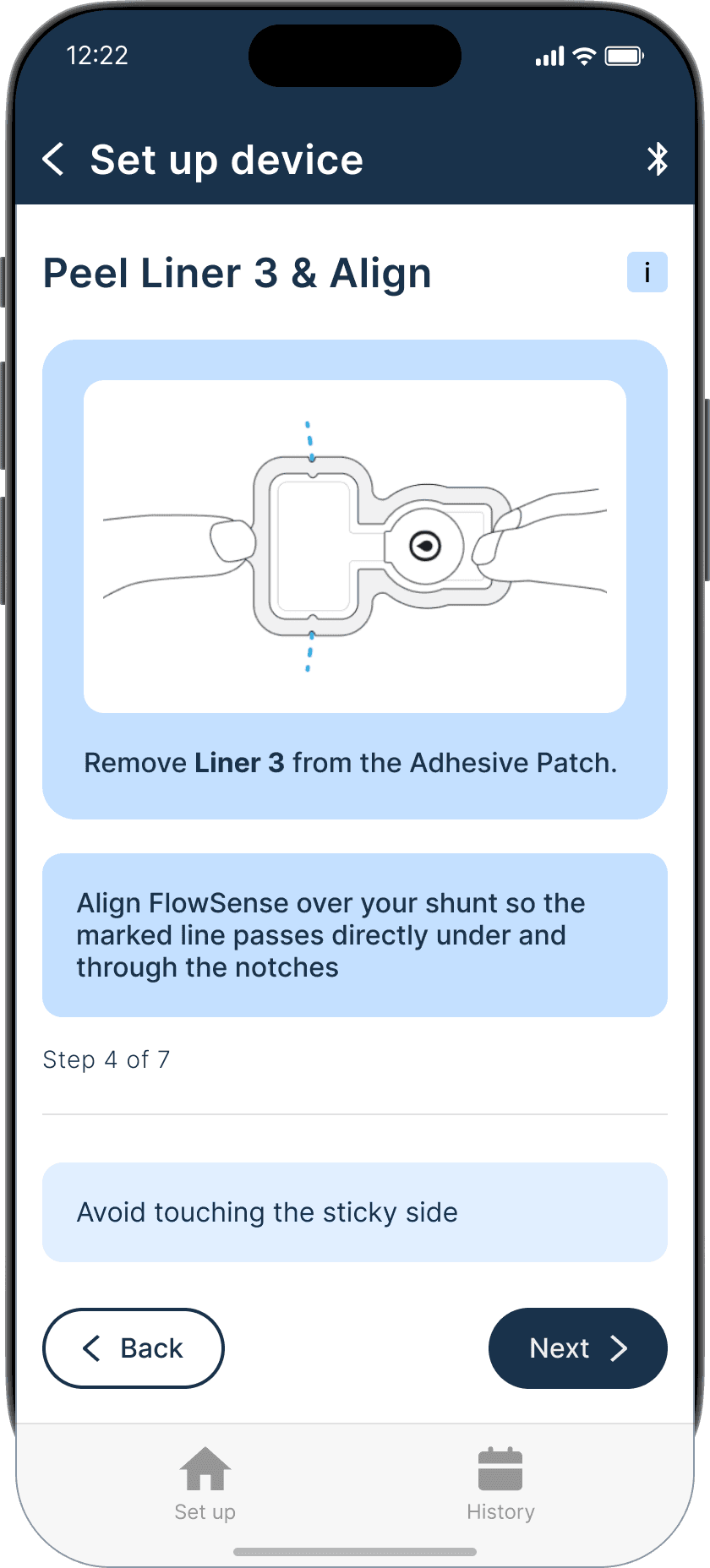
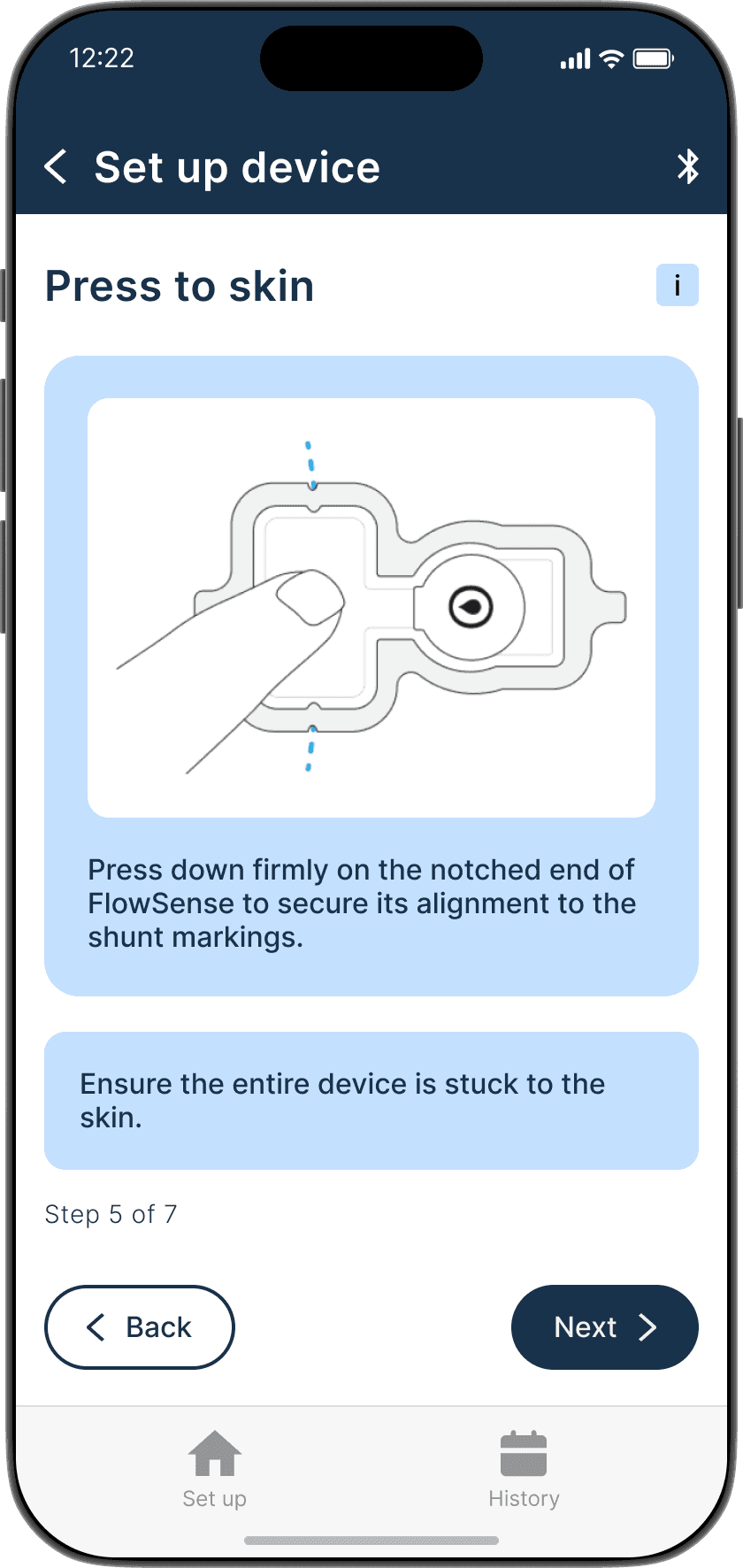
Applying the adhesive to the device & its alignment over the shunt is critical
Proper skin contact ensures accurate readings and reduced misplacement errors


Adhesive to the device & its alignment over the shunt is critical


Proper skin contact ensures accurate readings & reduced misplacement errors


Bluetooth pairing with FlowSense device




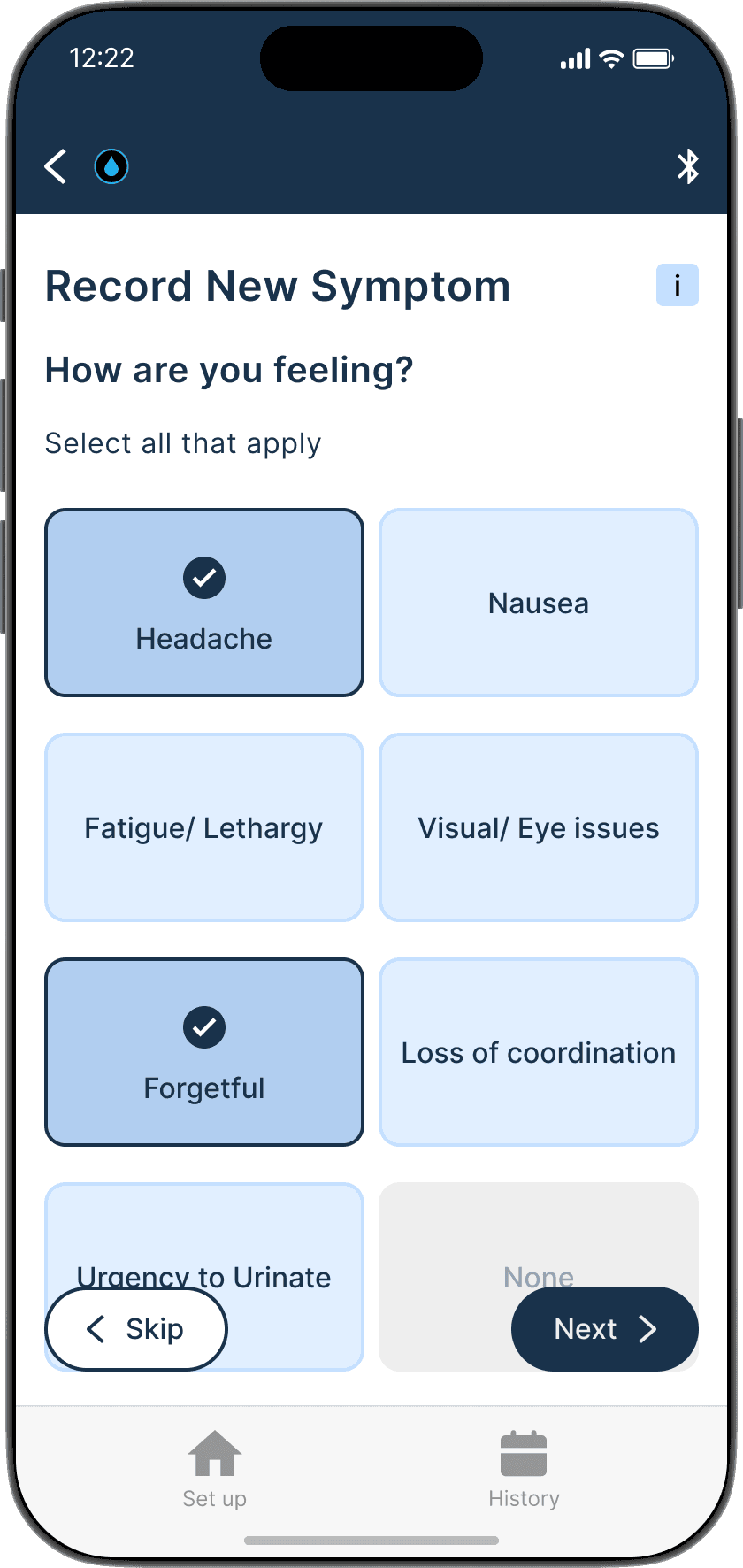
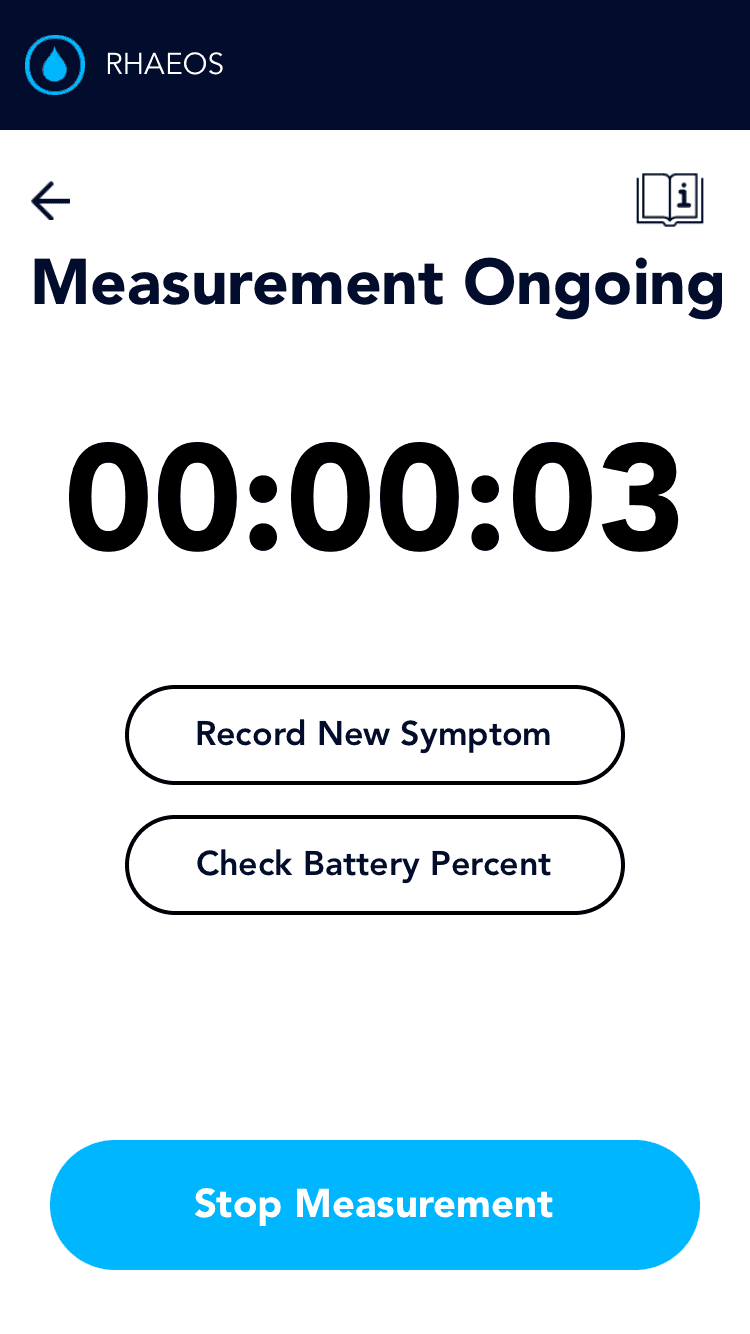
Symptom logging is crucial and provides context for measurement data, enabling more informed treatment decisions.
Symptom logging is crucial and provides context for measurement data, enabling more informed treatment decisions.
Symptom logging is crucial and provides context for measurement data, enabling more informed treatment decisions.
Accessibility Considerations
Accessibility Considerations


Allows patients to log symptoms before, during, and after the measurment
Allows patients to log symptoms before, during, and after the measurment


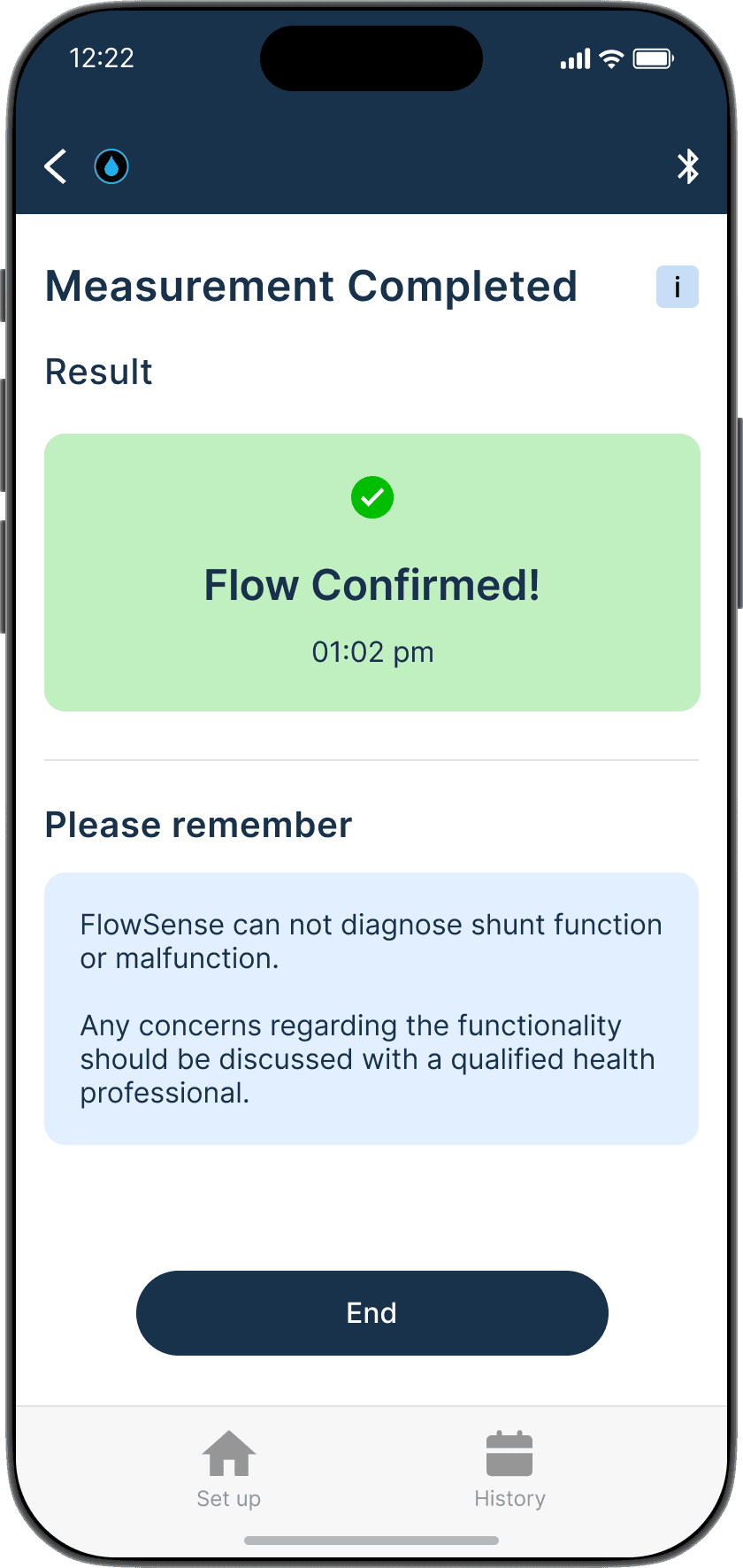

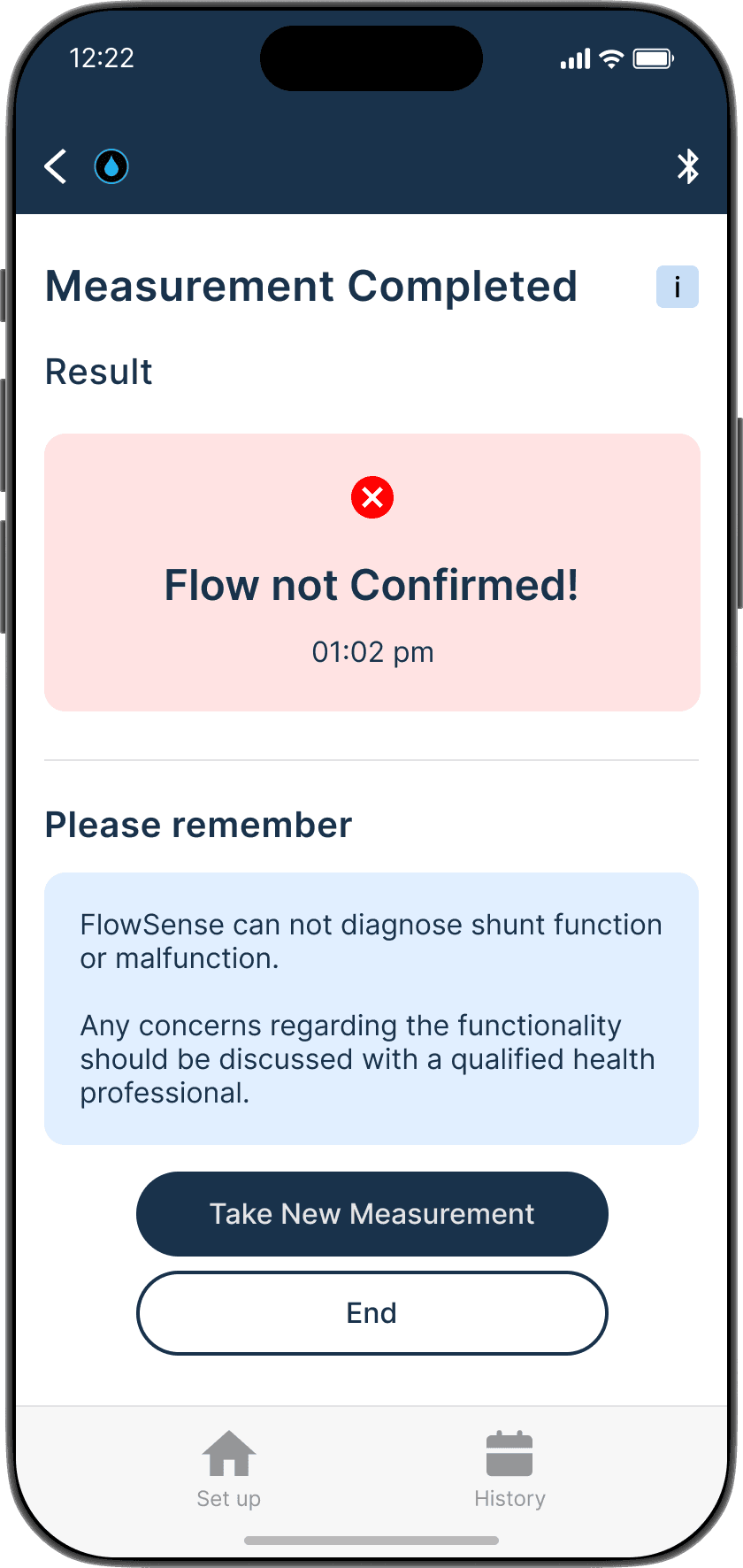

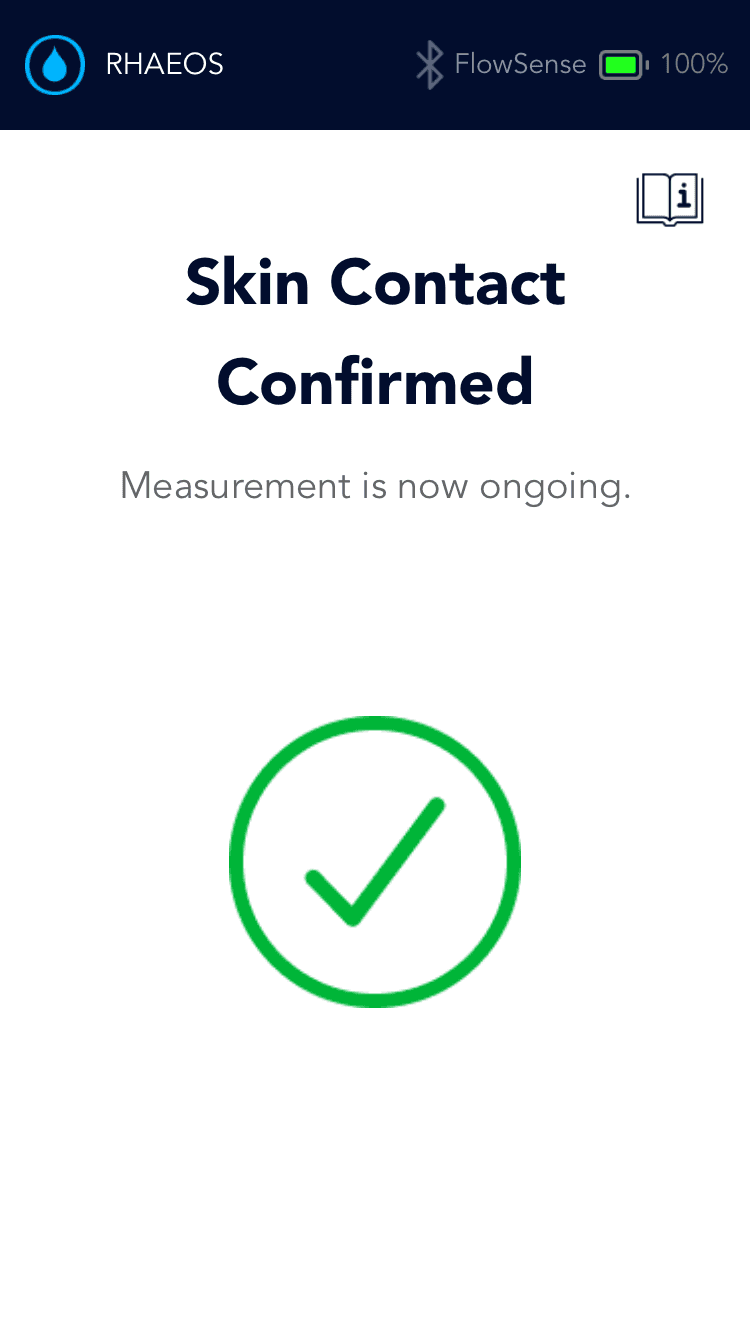
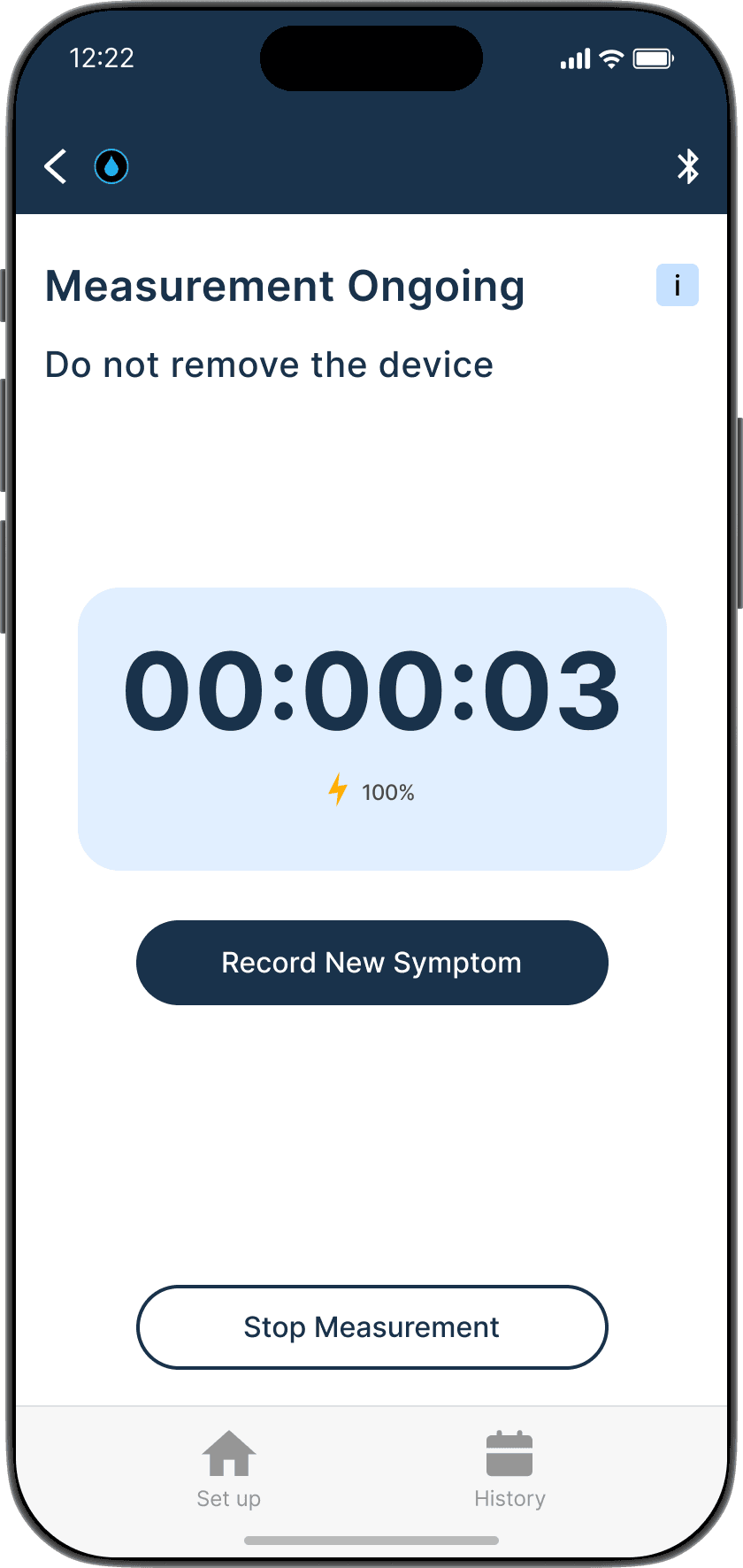
Ensures the device is properly adhered to the skin
Shunt flow successful
Shunt flow unsuccessful: is not always an emergency but good if tracked timely


Ensures the device is properly adhered to the skin


Flow successful


Flow unsuccessful: is not always an emergency but good if tracked timely

Phase 02: Pilot testing
Insights from Usability Testing & Patient Interviews from Pilot Study
Insights from Usability Testing & Patient Interviews from Pilot Study
Think-Aloud Protocol
(in person)
Task Analysis
(in person)
Observational Analysis
(in person)
Remote Usage Analytics
(session analystics, function engagement)
Semi Structured Interviews
(post trial, hybrid)
Think-Aloud Protocol
(in person)
Task Analysis
(in person)
Observational Analysis
(in person)
Semi Structured Interviews
(post trial, hybrid)
Remote Usage Analytics
(session analystics, function engagement)
Observational Analysis
(in person)
Semi Structured Interviews
(post trial, hybrid)
Phase 02: Pilot testing
Insights from Usability Testing & Patient Interviews from Pilot Study
Think-Aloud Protocol
(in person)
Task Analysis
(in person)
Observational Analysis
(in person)
Remote Usage Analytics
(session analystics, function engagement)
Semi Structured Interviews
(post trial, hybrid)
Think-Aloud Protocol
(in person)
Task Analysis
(in person)
Observational Analysis
(in person)
Semi Structured Interviews
(post trial, hybrid)
Remote Usage Analytics
(session analystics, function engagement)
Observational Analysis
(in person)
Semi Structured Interviews
(post trial, hybrid)
83%
83%

Patient satisfaction
Patient satisfaction
Patient satisfaction
"The app was so simple and easy to follow. I never felt lost at any step."
"The app was so simple and easy to follow. I never felt lost at any step."
"The app was so simple and easy to follow. I never felt lost at any step."
27%
27%

Suggested features
Suggested features
Suggested features
More troubleshooting tips
Real-time CSF flow visualization
More troubleshooting tips
Real-time CSF flow visualization
More troubleshooting tips
Real-time CSF flow visualization
3/72

Challenges
Challenges
3/72 users faced challenges
Device connectivity
Data upload error
3/72 users faced challenges
Device connectivity
Data upload error
3/72


Challenges
Challenges
3/72 users faced challenges
Device connectivity
Data upload error
Phase 02: Pilot testing
Insights from Usability Testing & Patient Interviews from Pilot Study
83%


Patient satisfaction
"The app was so simple and easy to follow. I never felt lost at any step."
27%


Suggested features
More troubleshooting tips
Real-time CSF flow visualization
3/72


Challenges
3/72 users faced challenges
Device connectivity
Data upload error
Process
Design Evolution
Method: Heuristic Evaluation